What is Magnesium Carbonate?
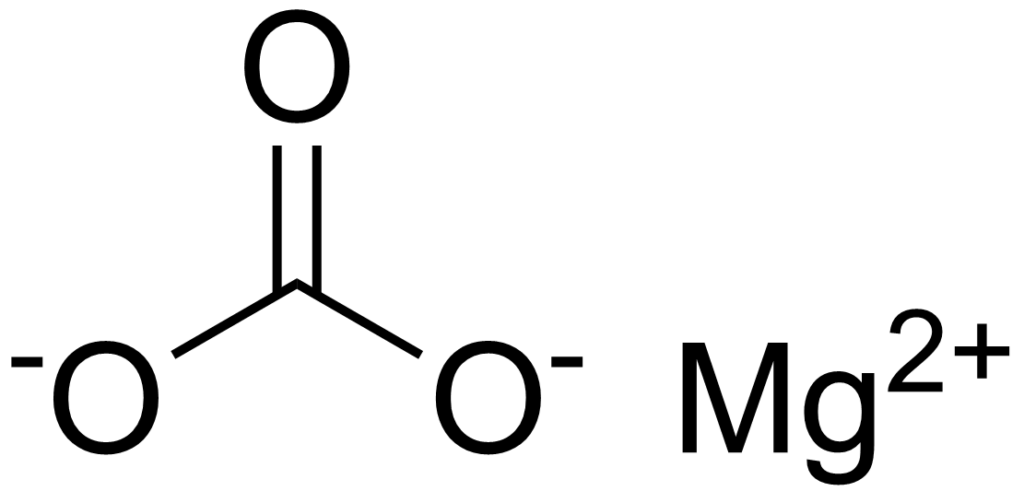
Magnesium carbonate is an inorganic compound of magnesium and carbonate ions with the chemical formula MgCO3. It’s a white to colorless solid mineral found naturally in certain rocks and minerals such as dolomite, magnesite, and limestone. It can also be produced synthetically by reacting soluble magnesium salt and sodium bicarbonate.
Also known as magnesite, it is a highly versatile mineral that performs various essential functions in foods and beverages. Relied on since the early 1900s, today, magnesium carbonate is used in a broad spectrum of products ranging from soda to bread to improve nutrition, texture, taste, and quality.
Common Forms of Magnesium Carbonate
Magnesium carbonate takes a variety of forms. The most common are the anhydrous salt called magnesite (MgCO3), and the di, tri, and pentahydrates known as barringtonite (MgCO3·2H2O), nesquehonite (MgCO3·3H2O), and lansfordite (MgCO3·5H2O), respectively.
Some basic forms, such as artinite (Mg2CO3(OH)2·3H2O), hydromagnesite (Mg5(CO3)4(OH)2·4H2O), and dypingite (Mg5(CO3)4(OH)2·5H2O), also occur as minerals.
Use of Magnesium Carbonate in Food and Beverage Products
Magnesium carbonate performs several valuable functions in foods and beverages. Here are several of the most popular uses of magnesium carbonate.
- Antacid
- Clarifying agent
- Acidity regulator
- Stabilizer
- Raising agent
- Anticaking agent
- Mineral source
- Dough modifier
- Emulsifier
- Adsorbent
- Laxative
- Colorant
Applications in Food and Beverage Products
Because magnesium carbonate is so versatile, it’s used in a variety of ways in many different types of foods and beverages. The following table illustrates common applications.
| Category | Products | Function |
|---|---|---|
| Bakery | Breads Doughs | Strengthening Agent Dough Conditioner Raising Agent Stabilizer |
| Beverages | Juices Soft Drinks | Acidity Regulator Carbonating Agent |
| Convenience Food | Cereals Powdered Beverages Ready-to-Cook Foods | Anticaking Agent Conditioner Emulsifier |
| Dairy | Ice Cream Yogurt | Stabilizer |
| Confectionery | Candies Chocolates | Color Additive Stabilizer |
| Others | Table Salt Table Sweeteners | Anticaking Agent Adsorbent |
Properties of Magnesium Carbonate
| Molecular Weight | 84.3139 g/mol |
| Melting Point | 350 °C decomposes (anhydrous) 165 °C (trihydrate) |
| Density | 2.958 g/cm3 |
| Storage Temperature | Room temperature at low humidity |
| Solubility in Water | Practically insoluble |
| Physical Form | Powder |
| Color | White |
| pH | 9.5 – 10.5 (5 % Aqueous solution) |
| Claims (*Product Specific) | Vegan, Gluten-free, Halal*, Kosher* |
Typical Formulations
Flavored Beverage
Beverage products are often enriched with calcium and magnesium in soluble form for nutritional purposes.
Here is an example of a beverage formulation table with Magnesium Carbonate along with the % weight of ingredients:
| Ingredient | % Composition |
|---|---|
| Calcium Carbonate | 2.5 g |
| Magnesium Carbonate | 1 g |
| Citric Acid | 8 g |
| Sugar | 100 g |
| Oligosaccharide | 15 g |
| Ascorbic Acid | 500 mg |
| Plant Extracts | 100 mg |
Bread
Magnesium carbonate is used in bread for nutrition, quality improvement, and taste enhancement.
Here is an example of a bread formulation table with Magnesium Carbonate along with the % weight of ingredients:
| Ingredient | Composition |
|---|---|
| Brew | |
| Water | 68 pounds |
| Salt | 9 ounces |
| Sugar | 11 ounces |
| Yeast | 2 pounds |
| Yeast Food | 8 ounces |
| Bread Dough | |
| Flour | 100 pounds |
| Sugar | 8 pounds |
| Salt | 6 ounces |
| Shortening (Vegetable Oil) | 3 pounds |
| Calcium Propionate | 4 ounces |
| Dough Developer | 2 pounds |
| Standard Enrichment Tablet (Vitamin B1, B2, B3 & Iron) | |
| Emulsifier | 1 pound |
| Magnesium Carbonate | 7.5 ounces |
Magnesium Carbonate Formulation Considerations
Magnesium carbonate’s properties create a variety of formulation implications. Here are seven considerations to take into account when using magnesium carbonate as an additive in food and beverage products.
Stability
Magnesium carbonate is light and oxidative stable. It’s heat stable up to 600°C.
Dosage
The typical dosage of magnesium carbonate in food products is between 0.1% and 0.5% by weight. However, dosage recommendations vary significantly depending on the function and application. Following optimal dosages is important since adding too much magnesium carbonate can negatively impact food quality. Breads are especially sensitive to magnesium carbonate, resulting in taste and texture problems and yeast inactivation (due to increased pH) when too much magnesium carbonate is added.
Effects on Textural Properties of Food Products
Magnesium carbonate has a binding or strengthening effect on gluten and adds strength to dough — adding magnesium carbonate to water changes how electrostatic forces act upon gluten, resulting in a change in dough rheology. The additional firmness imparted to the gluten by the salt enables it to better hold water and gas, allowing the dough to expand without tearing.
This influence becomes significant when soft water is used for dough mixing or when immature flour must be used. Also, due to its strong water-retaining capacity, magnesium carbonate dehydrates gluten, improving the machinability and handling of the dough.
Effects on Physicochemical Properties
Magnesium carbonate is an alkaline compound. As a result, it can act as an acidity regulator, neutralizing acids and raising the pH of food. For example, magnesium carbonate slows the fermentation rate by reducing the pH, checking yeast development, and preventing undesirable or excessive acidity. The shift in pH affects the physicochemical properties of food.
Magnesium carbonate also acts as a buffer by reacting with both acids and bases. It can absorb and neutralize acids, preventing them from affecting the pH of a solution. This helps to maintain a stable pH level in the solution. Magnesium carbonate decomposes to form magnesium oxide and carbon dioxide gas when heated.
Effects on Nutritional Properties
Magnesium is crucial to the health of the human body. It plays a role in muscle movement, bone strength, blood sugar levels, and cardiovascular health. Magnesium carbonate is a magnesium source, so foods fortified with magnesium carbonate can help increase magnesium intake.
Effects on Sensory Properties
Magnesium carbonate has an earthy, slightly bitter, salty taste. It is odorless — but since it has a high absorptive ability, it can absorb odors. Magnesium carbonate is typically white and will impart its color to food. For this reason, it’s also used as a whitening agent.
Interaction with Other Components
Magnesium carbonate can react with acids and bases to form salts or act as a leavening agent by reacting with weak acids to produce carbon dioxide gas. It can also react with other ingredients to form complex molecules affecting a food’s taste, texture, and stability.
Magnesium Carbonate Safety & Regulatory Considerations
Magnesium carbonate is safe for use in food when limited to recommended amounts. It’s allowed as a food additive in the United States and the European Union. The Food and Drug Administration (FDA) has established an acceptable daily intake of magnesium carbonate of up to 0.5 grams per kilogram body weight per day.
Identification Numbers
| CAS Number | 546-93-0 |
| EC Number | E504 |
Fun Facts About Magnesium Carbonate
- Magnesium carbonate is also used as an antacid and a fertilizer, among many other use cases.
- Because of its low solubility in water and hygroscopic properties, magnesium carbonate was first added to salt in 1911 to make it flow more freely.
Additional Sources & Resources
- NIH – Magnesium Carbonate
- Morton Salt – HER DEBUT
- Cargill – Salt in Bread Dough
- Google Patents – Calcium fortification of bread dough
- 1Source – E504; Magnesium Carbonate
- Magnesia – Perfect for food industry: Mineral compounds as helpful additives
- Wuxuwang – Magnesium Carbonate
- Journal of Food Science – EFFECT OF VARIOUS MAGNESIUM SOURCES ON BREADMAKING CHARACTERISTICS OF WHEAT FLOUR
- NIH – Effects of calcium–magnesium carbonate and calcium–magnesium hydroxide as supplemental sources of magnesium on microbial fermentation in a dual-flow continuous culture